What Was The Chemical Makeup Of The Atmosphere During The Trassic Period
Atmospheric composition of the Earth
The atmosphere of the World has been changing continuously since its formation. The Sun, the Earth and other planets were formed when matter coalesced from a rotating nebula approximately iv,567 million years ago, according to the most widely accepted theory.
The solar nebula originated from the explosion of older stars containing heavy elements like iron that had been created by nuclear fusion of lighter atoms. The accumulation of mass at the eye of the rotating nebula was so large that gravitational compression initiated the fusion of hydrogen into helium thus giving birth to the Lord's day which became a new shining star. The planets orbiting the Dominicus formed past accession; the heavier elements full-bodied in the cores and the lighter gaseous elements became the atmospheres.

Composition of the protoplanetary nebula.
The nigh abundant chemical elements in the Sun are hydrogen (H 73.46%), helium (He 24.85%), oxygen (O 0.77%), carbon (C 0.29%), iron (Iron 0.16%) and smaller percentages of neon, silicon, magnesium, sulfur, etc.[1] The atmosphere of Jupiter, which is the largest planet, is generally hydrogen with near ten% helium and modest amounts of other gases like methane, ammonia, hydrogen sulfide and water. These compositions indicate that the nebula from which our solar system originated was mostly hydrogen, helium and small-scale amounts of heavier elements. Planets like Mercury, Venus, Globe and Mars which are close to the Sun lost their hydrogen and helium speedily because their gravitational pull was not strong enough to retain these light elements. The loss of hydrogen and helium increased the concentration of heavier gases in the atmospheres of these terrestrial planets. Further away from the Sun where it is much colder, methyl hydride can condense as a liquid, and Saturn's moon Titan has a predominantly nitrogen atmosphere with pools of liquid methyl hydride on its surface.[xv]

The Earth's magnetosphere protects
the temper from the solar wind.

Aurora lite display around
the South Magnetic Pole
Earth's primordial atmosphere (Hadean Eon, 4.56 to iv.0 Ga)
When the material forming the Earth coalesced and melted, information technology organized itself into layers with dense materials at the cadre and less dumbo compounds closer to the surface. The gases comprising the atmosphere formed the outermost layer and had a composition similar to that of the gases of the condensing planetary nebula.
During the Hadean Eon, the Globe'due south surface consisted of molten rock, a magma ocean, and water existed only equally steam in the atmosphere. The circulation of molten metallic fe-nickel blend in the core of the Earth established the magnetosphere, a region in space where the motions of gas and fast charged particles are controlled by the magnetic field of the Earth. The magnetosphere deflects well-nigh solar wind ions before they penetrate the atmosphere, but the charged particles which are not deflected are directed toward the Earth's magnetic poles where high-energy collisions with atoms of the atmosphere produce an aurora light display. The average speed of hydrogen molecules and helium atoms is greater than the escape velocity from the Globe, and these calorie-free gases were lost and swept away through photoevaporation by the solar wind early in the Hadean Eon due to Earth's weak gravity.
Effectually iv.45 Ga, the Earth experienced a violent collision with a planetoid called Theia that was about the size of Mars. The affect added actress mass to the Earth, but a portion of the touch on droppings went into orbit and accreted to form the Moon.[10] Some scientists have proposed that this giant impact blasted away into space all of the Earth's atmosphere, including much of the water, and that the atmosphere and water were subsequently replenished past volcanic outgassing and impacts from asteroids and comets.[14] A problem with this proposal is that the ratio of deuterium to hydrogen (D/H) for comets is very dissimilar from what is found in the Earth'due south oceans, so comets are non a probable source for Earth's water. Also, volcanic emissions do not accept much nitrogen, then it is unlikely that volcanism provided the nitrogen in our electric current temper. While information technology is true that a slap-up standoff would have sent much of the atmosphere into infinite, most of information technology would have remained within the Earth'south gravitational sphere of influence and could accept been recaptured by the Earth equally the debris from the behemothic impact cooled and was partitioned between the World and the newly formed Moon giving both a similar chemic composition.
Afterward the hydrogen and helium had escaped, Earth's Hadean atmosphere was left with methane, ammonia, water vapor, and small percentages of nitrogen and carbon dioxide. A cataclysmic meteorite bombardment around 3.9 Ga kept much of the Earth's surface in the molten country, and the incoming impactors may have brought additional h2o, methane, ammonia, hydrogen sulfide and other gases that supplemented the atmosphere.
The high surface temperature of the Earth during the Hadean eon favored the depletion of atmospheric methane through the endothermic reaction of methane with the steam in the temper. Reactions such as the post-obit crave high temperatures of approximately 700°C to 1100°C which would have been common in the hot crust and magma lakes of the Hadean Earth. The resulting carbon monoxide would readily combine with metals to form carbonyl compounds.
CH4 + H2O![]() CO + 3 H2
CO + 3 H2
Methyl hydride reacts with steam to produce carbon monoxide and hydrogen.
The Hadean Eon was too hot for liquid water to condense on the surface of the Earth, only water vapor would have been able to condense at high altitude in the atmosphere and produce rain that evaporated rapidly as it savage when information technology approached the ground.
Toward the end of the Hadean Eon, volcanic activity started increasing the percentage of carbon dioxide in the temper. The Earth's surface inverse from molten lava to solid stone, and liquid water started to accumulate on the surface.
Globe'due south 2d atmosphere (Archean Eon, four.0 to 2.5 Ga)
The crust of the Earth started to cool downward during the Archean Eon. The amount of water vapor in the atmosphere decreased as h2o started condensing in liquid course. Continuous rainfall for millions of years led to the buildup of the oceans. As steam condensed into water, the atmospheric pressure of the World became lower, and the h2o started dissolving gases like ammonia and removed them from the atmosphere creating ammonium compounds, amines and other nitrogen-containing substances suitable for the origin of life. The condensation of water with gases such as sulfur dioxide produced acid rain that created new minerals on the Globe's surface. Volcanic carbon dioxide peaked during the Archean Eon and started to decrease through the germination of carbonate minerals that resulted from reactions of metals with the carbonic acid generated from carbon dioxide and water.
NH3 + H2O![]() NH4 + + OH-
NH4 + + OH-
COii + HtwoO![]() H2CO3
H2CO3
Reactions of ammonia and carbon dioxide with water

Calcite crystal consisting of calcium carbonate (CaCOiii)
Microfossils of sulfur-metabolizing cells take been found in three.4-billion-year-old rocks[six], and information technology is known that the first aquatic photosynthetic organisms originated around 3.5 Ga. The oxygen produced by cyanobacteria (blue-green algae) during the Archean Eon reacted with the metal ions in the anoxic sea. Billions of years would laissez passer before the photosynthetic microorganisms could eventually change the composition of the temper. By the middle of the Archean Eon, the Earth had cooled enough then that most of the water vapor in the atmosphere had condensed equally water, and the Earth had its beginning days without clouds. Ammonia and methane were but minor constituents of the temper. Carbon dioxide comprised nigh 15% of the atmosphere and the percentage of nitrogen was 75%.[5] In essence, most of the original components of the atmosphere had escaped, precipitated as liquids or reacted chemically to course solid compounds. The volcanic activity and the photosynthetic leaner were now the major factors influencing the Earth's atmospheric limerick.
Earth's third atmosphere (Proterozoic Eon, ii.5 to 0.54 Ga)
Monocellular life proliferated during the Proterozoic Eon. Anaerobic microbial life thrived in a planet with piddling oxygen. Anaerobic organisms obtained their energy in diverse ways. Methanogens combined hydrogen and carbon dioxide to produce methane and water:
CO2 + 4 Htwo ![]() CHfour + 2 H2O
CHfour + 2 H2O
Sulfate reducing bacteria combined methyl hydride and sulfate radicals:
CH4 + SO4 -- ![]() HCOthree - + HS- + H2O
HCOthree - + HS- + H2O
Other organisms capable of photosynthesis used the energy of sunlight to convert the abundant carbon dioxide and h2o into carbohydrates (C6H12O6) and oxygen, which was deadly to the anaerobes.
half-dozen COtwo + half dozen HiiO![]() Chalf-dozenH12Ohalf-dozen + 6 O2
Chalf-dozenH12Ohalf-dozen + 6 O2
Production of oxygen through photosynthesis
By the commencement quarter of the Proterozoic Eon, the Sun had become brighter and its luminosity had increased to 85% of the present level. By this time, near of the carbon dioxide had been depleted from the atmosphere, leaving nitrogen as the main atmospheric gas with a small pct of oxygen. Nitrogen gas (Nii ), which is quite inert chemically, had been a small-scale per centum of the Earth'south atmosphere during the Hadean Eon, simply it became the major component of the temper during the Proterozoic Eon in one case all the other gases were gone.


Banded Iron Germination (BIF) from Australia
The Globe's surface and seas contained slap-up quantities of fe that readily combined with oxygen to produce iron oxides. From the beginning of the Proterozoic Eon to ane.85 Ga, atmospheric oxygen levels rose as the rate of photosynthesis increased considerably. Shallow seas became partially oxygenated but the deep oceans continued to be anoxic. Although photosynthetic organisms had been releasing oxygen since Archean times, the oxygen levels could non build up in the atmosphere because the oxygen was being depleted by the oxidation of metals and by the oxidation of methane to yield carbon dioxide and water in the presence of ultraviolet (UV) radiation.
4 Fe + 3 Oii ![]() 2 Atomic number 262O3
2 Atomic number 262O3
Oxidation of metallic iron to course iron(III)oxide
CH4 + 2 Otwo ![]() CO2 + 2 H2O
CO2 + 2 H2O
Oxidation of methane
The cooling of the Earth during the Proterozoic stabilized the land masses and reduced the volcanic outgassing of carbon dioxide. Methyl hydride and carbon dioxide are greenhouse gases; their subtract in the atmosphere may have contributed to the Huronian glaciation that lasted from 2.4 Ga to two.i Ga. The common cold temperature sequestered additional methane from the temper by forming marsh gas clathrate, a crystal structure of water similar to ice that traps a big amount of methane. An increased period of oxygen production occurred between 2.4 Ga and 2.0 Ga and is known every bit the Great Oxidation Event or Oxygen Catastrophe. The higher oxygen level created banded iron formations (BIF) by precipitating dissolved atomic number 26.[vii] The reaction of oxygen with iron in its reduced country (Fe2+ ) continued to create BIF deposits of atomic number 26 in its oxidized state (Atomic number 263+ ) until about 1.9 Ga whenever volcanic action or crustal plate movements exposed unoxidized iron. Additional oxygen continued to be consumed past oxidation of minerals on the World's crust, but enough complimentary oxygen accumulated in the atmosphere to kill anaerobes near the Earth's surface thus creating an opportunity for the evolution of aerobic life forms.
Starting around 2.4 Ga, oxygen molecules migrated into the upper atmosphere and formed an ozone layer. This is a region in the stratosphere located betwixt 15 to 35 kilometers above the Earth'southward surface where oxygen molecules (Otwo ) are converted to ozone (Oiii ) by the Lord's day's ultraviolet rays. The contrary conversion of ozone back to oxygen releases oestrus. The ozone layer basically absorbs high-energy ultraviolet radiation and converts information technology to heat. The loftier free energy UV lite is unsafe for life because it can cause mutations in Dna sequences.
The atmospheric composition was very steady between i.85 Ga to 0.85 Ga. During this fourth dimension, Earth's atmosphere had approximately 10% oxygen. Photosynthetic organisms were still producing oxygen at a high rate, just the reaction of oxygen with dissolved minerals in the deep oceans and with rock and clay on the World's surface did not allow atmospheric oxygen levels to increase. Past 0.85 Ga, the minerals in the ocean and on country could not bind as much oxygen, and the excess oxygen began to accumulate in the temper. With the increased oxygen levels and the protection of the ozone layer, organisms capable of aerobic respiration could at present proliferate all over the surface of the Earth.
Earth's present atmosphere (Phanerozoic Eon, 0.542 Ga to present)
The beginning of the Phanerozoic Eon, the Cambrian period, is marked past an abundance of multicellular life. Well-nigh of the major groups of animals first appeared at this time. Vegetation covered the surface of the Globe, and oxygen accounted for 30% of the atmosphere. Air enriched with so much oxygen allowed giant insects to develop and caused frequent wood fires ready off past lightning.

Meganeura, a dragonfly from the Carboniferous period had a wingspan of 65 cm (two.one ft)
A great mass-extinction effect occurred 251 one thousand thousand years ago (0.251 Ga) marking the boundary of the Permian and Triassic periods. Oxygen levels dropped from 30% to 12%, and carbon dioxide levels reached about 2000 ppm. This was Earth'southward worst mass extinction and it eliminated 90% of sea dwellers and 70% of land plants and animals. The crusade of this mass extinction is thought to take been a series of volcanic events in Siberia that lasted for about i 1000000 years and released large volumes of carbon dioxide and gases containing sulfur, chlorine and fluorine.[viii,12] By 228 million years ago, oxygen levels had risen to about 15% of the atmosphere, and the first dinosaurs appeared.[xi] Oxygen levels continued to increment, and by the finish‐Cretaceous, 100 million years ago, oxygen had risen to most 23% of the temper. At this time, dinosaurs were well established and modern mammals and birds began to develop. For the last 100 million years, the percentage of oxygen has fluctuated betwixt 18% and 23% to the nowadays level of almost 21% of the temper.[13] The post-obit table shows the percentages of the five most common gases in today'due south atmosphere.[9]
Principal components of World's atmosphere past volume
| Nitrogen (N2) | 780,840 ppm (78.084%) |
| Oxygen (O2) | 209,460 ppm (20.946%) |
| Argon (Ar) | 9,340 ppm (0.9340%) |
| Carbon dioxide (CO2) | 394.45 ppm (0.039445%) |
| Neon (Ne) | xviii.18 ppm (0.001818%) |
Earth'due south future atmosphere.
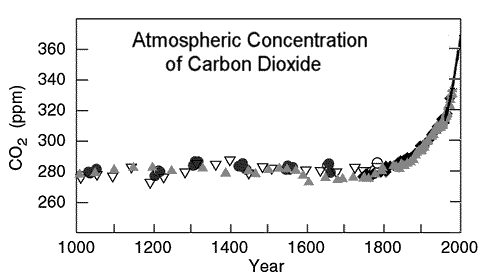
Since the get-go of the industrial revolution in 1750, humans accept been called-for coal and petroleum products to provide the energy to power machinery. The combustion of fossil fuels has been generating large quantities of the greenhouse gases carbon dioxide (CO2), methane (CH4), and nitrous oxide (N2O). The nautical chart below shows the levels of atmospheric carbon dioxide during the terminal millennium and the precipitous rise during the twentieth century.[2] The concentration of atmospheric carbon dioxide today is approximately 400 parts per one thousand thousand (ppm) and the North Pole's mean annual temperature is -xx°C. When the carbon dioxide concentration was 2,000 ppm 55 million years ago during the Paleocene–Eocene Thermal Maximum (PETM), the N Pole'due south temperature averaged 23°C (73.4°F).[3] It is thought that elevated levels of greenhouse gases volition crusade global warming and influence weather condition patterns. Many cities bordering the coastal areas will be permanently flooded if the water ice deposits in Greenland and Antarctica melt.
© Copyright - Antonio Zamora
Source: https://www.scientificpsychic.com/etc/timeline/atmosphere-composition.html
Posted by: millikenanturing.blogspot.com

0 Response to "What Was The Chemical Makeup Of The Atmosphere During The Trassic Period"
Post a Comment