What Is The Chemical Makeup Of A Person
Trunk composition may be analyzed in various ways. This can be done in terms of the chemical elements present, or by molecular blazon e.g., water, protein, fats (or lipids), hydroxylapatite (in basic), carbohydrates (such every bit glycogen and glucose) and Deoxyribonucleic acid. In terms of tissue type, the body may be analyzed into water, fat, connective tissue, muscle, bone, etc. In terms of jail cell blazon, the body contains hundreds of different types of cells, but notably, the largest number of cells contained in a homo body (though not the largest mass of cells) are not human cells, but bacteria residing in the normal human gastrointestinal tract.
Elements [edit]
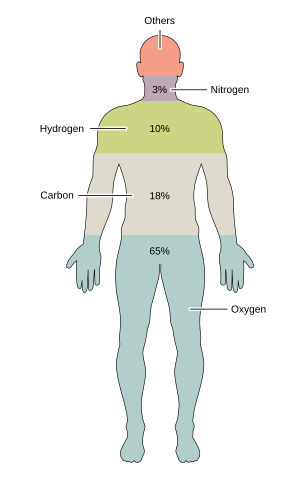 | Element | Symbol | percent mass | percent atoms |
|---|---|---|---|---|
| Oxygen | O | 65.0 | 24.0 | |
| Carbon | C | 18.5 | 12.0 | |
| Hydrogen | H | 10 | 62.0 | |
| Nitrogen | Due north | iii.2 | 1.1 | |
| Calcium | Ca | one.5 | 0.22 | |
| Phosphorus | P | ane.0 | 0.22 | |
| Potassium | K | 0.4 | 0.03 | |
| Sulfur | South | 0.iii | 0.038 | |
| Sodium | Na | 0.2 | 0.037 | |
| Chlorine | Cl | 0.ii | 0.024 | |
| Magnesium | Mg | 0.ane | 0.015 | |
| All others | < 0.1 | < 0.three |

Pie charts of typical human torso composition by percent of mass, and past percent of atomic composition (atomic pct).
Virtually 99% of the mass of the human torso is made up of six elements: oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. Only about 0.85% is composed of some other 5 elements: potassium, sulfur, sodium, chlorine, and magnesium. All 11 are necessary for life. The remaining elements are trace elements, of which more than than a dozen are thought on the basis of expert evidence to be necessary for life.[i] All of the mass of the trace elements put together (less than x grams for a homo body) exercise not add up to the torso mass of magnesium, the least common of the xi not-trace elements.
Other elements [edit]
Not all elements which are found in the human body in trace quantities play a office in life. Some of these elements are thought to exist unproblematic common contaminants without function (examples: caesium, titanium), while many others are thought to be active toxins, depending on amount (cadmium, mercury, atomic number 82, radioactives). In humans, arsenic is toxic, and its levels in foods and dietary supplements are closely monitored to reduce or eliminate its intake.[ii]
Some elements (silicon, boron, nickel, vanadium) are probably needed by mammals too, but in far smaller doses. Bromine is used abundantly by some (though not all) lower[ clarification needed ] organisms, and opportunistically in eosinophils in humans. Ane report has indicated bromine to be necessary to collagen IV synthesis in humans.[3] Fluorine is used past a number of plants to manufacture toxins merely only functions in humans as a local topical hardening amanuensis in tooth enamel.[four]
Elemental composition list [edit]
The average 70 kg (150 lb) adult man body contains approximately 7×1027 atoms and contains at to the lowest degree detectable traces of 60 chemic elements.[5] About 29 of these elements are thought to play an active positive role in life and health in humans.[six]
The relative amounts of each chemical element vary by individual, mainly due to differences in the proportion of fatty, musculus and bone in their torso. Persons with more fat will have a higher proportion of carbon and a lower proportion of most other elements (the proportion of hydrogen volition be about the same). The numbers in the table are averages of dissimilar numbers reported by different references.
The adult human being torso averages ~53% h2o.[seven] This varies substantially past historic period, sex, and adiposity. In a large sample of adults of all ages and both sexes, the effigy for water fraction by weight was institute to exist 48 ±half-dozen% for females and 58 ±eight% h2o for males.[8] H2o is ~11% hydrogen by mass just ~67% hydrogen by atomic percent, and these numbers along with the complementary % numbers for oxygen in water, are the largest contributors to overall mass and diminutive composition figures. Because of water content, the human body contains more oxygen by mass than any other chemical element, but more hydrogen past atom-fraction than whatsoever chemical element.
The elements listed below as "Essential in humans" are those listed by the (Us) Food and Drug Administration as essential nutrients,[9] likewise as six additional elements: oxygen, carbon, hydrogen, and nitrogen (the fundamental building blocks of life on Earth), sulfur (essential to all cells) and cobalt (a necessary component of vitamin B12). Elements listed as "Perchance" or "Probably" essential are those cited past the National Research Council (Us) as beneficial to homo health and possibly or probably essential.[x]
| Diminutive number | Element | Fraction of mass [11] [12] [13] [14] [xv] [16] | Mass (kg)[17] | Diminutive pct | Essential in humans[eighteen] | Negative effects of excess | Group |
|---|---|---|---|---|---|---|---|
| 8 | Oxygen | 0.65 | 43 | 24 | Yes (e.g. water, electron acceptor)[19] | Reactive oxygen species | 16 |
| half-dozen | Carbon | 0.18 | 16 | 12 | Yes[xix] (organic compounds) | xiv | |
| 1 | Hydrogen | 0.x | 7 | 62 | Yes[19] (due east.g. water) | Acidosis | one |
| 7 | Nitrogen | 0.03 | 1.8 | 1.1 | Yes[19] (e.g. Dna and amino acids) | 15 | |
| twenty | Calcium | 0.014 | 1.0 | 0.22 | Yes[nineteen] [20] [21] (e.g. Calmodulin and Hydroxylapatite in bones) | Hypercalcaemia | 2 |
| 15 | Phosphorus | 0.011 | 0.78 | 0.22 | Yes[19] [20] [21] (e.1000. Dna, Phospholipids and Phosphorylation) | Hyperphosphatemia | 15 |
| 19 | Potassium | 2.0×10−3 | 0.14 | 0.033 | Yes[xix] [20] (east.k. Na+/K+-ATPase) | Hyperkalemia | ane |
| 16 | Sulfur | ii.5×10−3 | 0.14 | 0.038 | Yeah[19] (e.g. Cysteine, Methionine, Biotin, Thiamine) | 16 | |
| 11 | Sodium | ane.5×10−3 | 0.10 | 0.037 | Yes[20] (eastward.g. Na+/K+-ATPase) | Hypernatremia | 1 |
| 17 | Chlorine | i.5×10−3 | 0.095 | 0.024 | Yes[20] [21] (eastward.thousand. Cl-transporting ATPase) | Hyperchloremia | 17 |
| 12 | Magnesium | 500×ten−6 | 0.019 | 0.0070 | Yes[20] [21] (e.m. binding to ATP and other nucleotides) | Hypermagnesemia | 2 |
| 26 | Iron* | 60×10−6 | 0.0042 | 0.00067 | Yes[20] [21] (e.k. Hemoglobin, Cytochromes) | Iron overload | 8 |
| 9 | Fluorine | 37×10−6 | 0.0026 | 0.0012 | Yes (AUS, NZ),[22] No (United states, EU),[23] [24] Possibly (WHO)[25] | Fluorine: Highly toxic Fluoride: Toxic in loftier amounts | 17 |
| 30 | Zinc | 32×10−half dozen | 0.0023 | 0.00031 | Yes[xx] [21] (e.m. Zinc finger proteins) | Zinc toxicity | 12 |
| 14 | Silicon | xx×10−vi | 0.0010 | 0.0058 | Probably[26] | xiv | |
| 31 | Gallium | 4.9×10−6 | 0.0007 | 0.00093 | No | Gallium halide poisoning[27] | 13 |
| 37 | Rubidium | four.6×10−6 | 0.00068 | 0.000033 | No | Potassium replacement | 1 |
| 38 | Strontium | 4.6×10−6 | 0.00032 | 0.000033 | —— | Calcium replacement | ii |
| 35 | Bromine | 2.nine×10−6 | 0.00026 | 0.000030 | —— | Bromism | 17 |
| 82 | Lead | 1.7×10−half-dozen | 0.00012 | 0.0000045 | No | Lead poisoning | fourteen |
| 29 | Copper | 1×10−6 | 0.000072 | 0.0000104 | Yes[20] [21] (due east.g. copper proteins) | Copper toxicity | eleven |
| 13 | Aluminium | 870×10−9 | 0.000060 | 0.000015 | No | Aluminium poisoning | 13 |
| 48 | Cadmium | 720×10−nine | 0.000050 | 0.0000045 | No | Cadmium poisoning | 12 |
| 58 | Cerium | 570×10−nine | 0.000040 | No | |||
| 56 | Barium | 310×x−ix | 0.000022 | 0.0000012 | No | toxic in higher amounts | two |
| fifty | Tin | 240×x−9 | 0.000020 | 6.0×10−7 | No | 14 | |
| 53 | Iodine | 160×x−9 | 0.000020 | 7.5×10−7 | Yes[twenty] [21] (e.thou. thyroxine, triiodothyronine) | Iodine-induced Hyperthyroidism[28] | 17 |
| 22 | Titanium | 130×10−9 | 0.000020 | No | 4 | ||
| 5 | Boron | 690×10−9 | 0.000018 | 0.0000030 | Probably[ten] [29] | thirteen | |
| 34 | Selenium | 190×ten−9 | 0.000015 | four.5×10−eight | Yes[twenty] [21] (e.k. selenocysteine) | Selenium toxicity | 16 |
| 28 | Nickel | 140×10−9 | 0.000015 | 0.0000015 | No | toxic | x |
| 24 | Chromium | 24×x−nine | 0.000014 | 8.ix×10−eight | Yes[20] [21] | vi | |
| 25 | Manganese | 170×10−9 | 0.000012 | 0.0000015 | Yes[20] [21] (east.m. Mn-SOD) | 7 | |
| 33 | Arsenic | 260×x−nine | 0.000007 | 8.9×10−eight | No[2] | Arsenic poisoning | 15 |
| iii | Lithium | 31×10−9 | 0.000007 | 0.0000015 | Yes (intercorrelated with the functions of several enzymes, hormones and vitamins) | Lithium toxicity | ane |
| 80 | Mercury | 190×ten−9 | 0.000006 | 8.9×x−8 | No | Mercury poisoning | 12 |
| 55 | Caesium | 21×10−nine | 0.000006 | ane.0×10−7 | No | one | |
| 42 | Molybdenum | 130×10−9 | 0.000005 | iv.5×10−8 | Yes[20] [21] (east.1000. the molybdenum oxotransferases, Xanthine oxidase and Sulfite oxidase) | vi | |
| 32 | Germanium | 5×x−six | No | 14 | |||
| 27 | Cobalt | 21×10−nine | 0.000003 | three.0×10−7 | Yes (e.grand. Cobalamin/Vitamin B12)[30] [31] | 9 | |
| 44 | Ruthenium | 22×10−9 | 0.000007 | No [32] | 8 | ||
| 51 | Antimony | 110×10−ix | 0.000002 | No | toxic | xv | |
| 47 | Silver | 10×x−9 | 0.000002 | No | 11 | ||
| 41 | Niobium | 1600×ten−9 | 0.0000015 | No | 5 | ||
| 40 | Zirconium | half-dozen×10−6 | 0.000001 | 3.0×10−vii | No | iv | |
| 57 | Lanthanum | 1370×10−9 | 8×10−7 | No | |||
| 52 | Tellurium | 120×10−9 | seven×ten−7 | No | 16 | ||
| 39 | Yttrium | 6×x−7 | No | 3 | |||
| 83 | Bismuth | five×10−7 | No | xv | |||
| 81 | Thallium | 5×10−7 | No | highly toxic | 13 | ||
| 49 | Indium | 4×ten−7 | No | 13 | |||
| 79 | Gold | 3×10−nine | two×10−7 | 3.0×10−7 | No | uncoated nanoparticles peradventure genotoxic[33] [34] [35] | 11 |
| 21 | Scandium | 2×ten−7 | No | 3 | |||
| 73 | Tantalum | two×10−seven | No | five | |||
| 23 | Vanadium | 260×10−9 | 1.i×10−seven | one.ii×10−8 | Peradventure[ten] (suggested osteo-metabolism (bone) growth factor) | 5 | |
| ninety | Thorium | 1×ten−7 | No | toxic, radioactive | |||
| 92 | Uranium | i×10−vii | 3.0×x−nine | No | toxic, radioactive | ||
| 62 | Samarium | five.0×10−viii | No | ||||
| 74 | Tungsten | 2.0×ten−8 | No | half-dozen | |||
| 4 | Beryllium | 3.half-dozen×10−8 | iv.5×x−eight | No | toxic in higher amounts | ii | |
| 88 | Radium | iii×10−xiv | 1×ten−17 | No | toxic, radioactive | 2 |
*Iron = ~3 grand in males, ~2.3 g in females
Of the 94 naturally occurring chemic elements, 61 are listed in the table above. Of the remaining 33, it is non known how many occur in the human body.
Most of the elements needed for life are relatively common in the World's crust. Aluminium, the tertiary most common element in the Earth'south crust (after oxygen and silicon), serves no function in living cells, but is toxic in big amounts, depending on its physical and chemical forms and magnitude, duration, frequency of exposure, and how it was absorbed past the human being body.[36] Transferrins can demark aluminium.[37]
Periodic tabular array [edit]
| Nutritional elements in the periodic table[38] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H | He | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thousand | Ca | Sc | Ti | Five | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | Every bit | Se | Br | Kr | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cs | Ba | * | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fr | Ra | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ** | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fable:
Quantity elements
Deemed essential trace element past U.South., not by European Spousal relationship
Suggested role from deprivation effects or active metabolic handling, but no clearly-identified biochemical function in humans
Limited coexisting evidence for trace benefits or biological action in mammals
No evidence for biological activity in mammals, simply essential in some lower organisms.
(In the example of lanthanum, the definition of an essential food as being indispensable and irreplaceable is not completely applicative due to the extreme similarity of the lanthanides. The stable early lanthanides up to Sm are known to stimulate the growth of various lanthanide-using organisms.)[39]
Molecules [edit]
The composition of the man body expressed in terms of chemicals:
- Water
- Proteins – including those of hair, connective tissue, etc.
- Fats (or lipids)
- Hydroxyapatite in bones
- Carbohydrates such every bit glycogen and glucose
- DNA
- Dissolved inorganic ions such as sodium, potassium, chloride, bicarbonate, phosphate
- Gases such as oxygen, carbon dioxide, nitrogen oxide, hydrogen, carbon monoxide, acetaldehyde, formaldehyde, methanethiol. These may be dissolved or nowadays in the gases in the lungs or intestines. Ethane and pentane are produced by oxygen free radicals.[twoscore]
- Many other pocket-sized molecules, such as amino acids, fatty acids, nucleobases, nucleosides, nucleotides, vitamins, cofactors.
- Gratis radicals such as superoxide, hydroxyl, and hydroperoxyl.
The limerick of the human body can be viewed on an atomic and molecular scale as shown in this article.
The estimated gross molecular contents of a typical 20-micrometre human cell is equally follows:[41]
| Molecule | Percent of mass | Mol. weight (daltons) | Molecules | Per centum of molecules |
|---|---|---|---|---|
| Water | 65 | 18 | i.74×xxiv | 98.73 |
| Other inorganics | 1.5 | N/A | ane.31×x12 | 0.74 |
| Lipids | 12 | N/A | 8.4×1011 | 0.475 |
| Other organics | 0.four | Northward/A | 7.7×10x | 0.044 |
| Protein | 20 | N/A | i.9×10x | 0.011 |
| RNA | 1.0 | N/A | 5×10seven | iii×10−5 |
| DNA | 0.1 | 1×ten11 | 46 | three×10−xi |
Tissues [edit]
 | Jail cell type | % mass | % cell count |
|---|---|---|---|
| Erythrocytes (red blood cells) | 4.two | 85.0 | |
| Muscle cells | 28.6 | 0.001 | |
| Adipocytes (fat cells) | 18.6 | 0.2 | |
| Other cells | 14.three | fourteen.8 | |
| Extracellular components | 34.3 | - |
Trunk composition can as well be expressed in terms of various types of material, such as:
- Muscle
- Fat
- Bone and teeth
- Nervous tissue (Encephalon and nerves)
- Hormones
- Connective tissue
- Body fluids (claret, lymph, Urine)
- Contents of digestive tract, including abdominal gas
- Air in lungs
- Epithelium
Limerick by cell type [edit]
At that place are many species of bacteria and other microorganisms that live on or inside the healthy man body. In fact, there are roughly as many microbial as homo cells in the human body past number.[43] [44] [45] [46] (much less by mass or volume). Some of these symbionts are necessary for our health. Those that neither aid nor harm humans are called commensal organisms.
Run into also [edit]
- List of organs of the human body
- Hydrostatic weighing
- Dietary element
- Composition of claret
- List of human being claret components
- Body composition
- Abundance of elements in Earth's crust
- Abundance of the chemical elements
References [edit]
- ^ M.A. Zoroddu; J. Aashet; G. Crisponi; S. Medici; Grand. Peana; 5.M. Nurchi (June 2019). "The essential metals for humans: a brief overview". Periodical of Inorganic Biochemistry. 195: 120–129. doi:10.1016/j.jinorgbio.2019.03.013. PMID 30939379. S2CID 92997696.
- ^ a b "Arsenic in Food and Dietary Supplements". US Food and Drug Administration. 22 May 2019. Retrieved 20 August 2019.
- ^ McCall AS, Cummings CF, Bhave Yard, Vanacore R, Folio-McCaw A, Hudson BG (2014). "Bromine Is an Essential Trace Element for Assembly of Collagen Iv Scaffolds in Tissue Development and Architecture". Jail cell. 157 (6): 1380–92. doi:10.1016/j.jail cell.2014.05.009. PMC4144415. PMID 24906154.
- ^ Nelson, Lehninger, Cox (2008). Lehninger Principles of Biochemistry (5th ed.). Macmillan.
{{cite volume}}: CS1 maint: multiple names: authors list (link) - ^ How many atoms are in the human being body?
- ^ "Ultratrace minerals". Authors: Nielsen, Forrest H. USDA, ARS Source: Modernistic nutrition in health and illness / editors, Maurice E. Shils ... et al.. Baltimore : Williams & Wilkins, c. 1999, p. 283-303. Issue Date: 1999 URI: [ane]
- ^ Utilize WP:CALC for the mean of means for males and females, since the ii groups are of about equal size
- ^ Meet table 1. here
- ^ "Guidance for Manufacture: A Food Labeling Guide 14. Appendix F". US Nutrient and Drug Assistants. 1 January 2013. Archived from the original on iv April 2017.
- ^ a b c Institute of Medicine (29 September 2006). Dietary Reference Intakes: The Essential Guide to Nutrient Requirements. National Academies Press. pp. 313–nineteen, 415–22. ISBN978-0-309-15742-1 . Retrieved 21 June 2016.
- ^ Thomas J. Glover, comp., Pocket Ref, tertiary ed. (Littleton: Sequoia, 2003), p. 324 (LCCN 2002-91021), which in
- ^ plow cites Geigy Scientific Tables, Ciba-Geigy Limited, Basel, Switzerland, 1984.
- ^ Chang, Raymond (2007). Chemistry, Ninth Edition. McGraw-Colina. p. 52. ISBN978-0-07-110595-eight.
- ^ "Elemental Limerick of the Human Torso" Archived 2018-12-18 at the Wayback Motorcar by Ed Uthman, MD Retrieved 17 June 2016
- ^ Frausto Da Silva, J. J. R; Williams, R. J. P (2001-08-xvi). The Biological Chemical science of the Elements: The Inorganic Chemistry of Life. ISBN9780198508489.
- ^ Zumdahl, Steven S. and Susan A. (2000). Chemistry, Fifth Edition. Houghton Mifflin Company. p. 894. ISBN978-0-395-98581-vi. )
- ^ Emsley, John (25 August 2011). Nature'southward Building Blocks: An A-Z Guide to the Elements. OUP Oxford. p. 83. ISBN978-0-19-960563-7 . Retrieved 17 June 2016.
- ^ Neilsen, cited
- ^ a b c d eastward f yard h Salm, Sarah; Allen, Deborah; Nester, Eugene; Anderson, Denise (9 Jan 2015). Nester'south Microbiology: A Man Perspective. p. 21. ISBN978-0-07-773093-ii . Retrieved 19 June 2016.
- ^ a b c d e f g h i j grand l grand n Subcommittee on the Tenth Edition of the Recommended Dietary Allowances, Nutrient and Diet Board; Commission on Life Sciences, National Research Council (1 February 1989). "9-10". Recommended Dietary Allowances: 10th Edition . National Academies Press. ISBN978-0-309-04633-6 . Retrieved 18 June 2016.
- ^ a b c d e f k h i j k fifty Code of Federal Regulations, Championship 21: Food and Drugs, Ch one, subchapter B, Part 101, Subpart A, §101.9(c)(8)(iv)
- ^ Australian National Health and Medical Research Council (NHMRC) and New Zealand Ministry of Health (MoH)
- ^ "Fluoride in Drinking Water: A Review of Fluoridation and Regulation Issues"
- ^ "Scientific Opinion on Dietary Reference Values for fluoride" (PDF). EFSA Journal. eleven (8): 3332. 2013. doi:ten.2903/j.efsa.2013.3332. ISSN 1831-4732.
- ^ WHO/SDE/WSH/03.04/96 "Fluoride in Drinking-water"
- ^ Muhammad Ansar Farooq; Karl-Josef Dietz (2015). "Silicon as Versatile Histrion in Found and Human Biology: Overlooked and Poorly Understood Muhammad Ansar Farooq and Karl-J". Front. Institute Sci. 6 (994): 994. doi:10.3389/fpls.2015.00994. PMC4641902. PMID 26617630.
- ^ Ivanoff, C. Due south.; Ivanoff, A. Due east.; Hottel, T. 50. (February 2012). "Gallium poisoning: a rare case report". Food Chem. Toxicol. 50 (2): 212–5. doi:ten.1016/j.fct.2011.10.041. PMID 22024274.
- ^ Healthline: Everything You Demand to Know About Iodine Poisoning
- ^ Safe Upper Levels for Vitamins and Mineral (2003), boron p. 164-71, nickel p. 225-31, EVM, Food Standards Agency, Britain ISBN 1-904026-xi-7
- ^ Yamada, Kazuhiro (2013). "Cobalt: Its Role in Health and Disease". Interrelations betwixt Essential Metal Ions and Human Diseases. Metal Ions in Life Sciences. Vol. 13. pp. 295–320. doi:10.1007/978-94-007-7500-8_9. ISBN978-94-007-7499-5. ISSN 1559-0836. PMID 24470095.
- ^ Banci, Lucia (18 April 2013). Metallomics and the Cell. Springer Science & Business Media. pp. 333–368. ISBN978-94-007-5561-1 . Retrieved 19 June 2016.
- ^ Toeniskoetter, Steve (2020). "Ruthenium". Biochemical Periodic Table.
- ^ Fratoddi, Ilaria; Venditti, Iole; Cametti, Cesare; Russo, Maria Vittoria (2015). "How toxic are gold nanoparticles? The state-of-the-art". Nano Research. 8 (half-dozen): 1771–1799. doi:ten.1007/s12274-014-0697-3. hdl:11573/780610. ISSN 1998-0124. S2CID 84837060.
- ^ "Scientific Opinion on the re-evaluation of gold (E 175) every bit a food additive". EFSA Journal. 14 (1): 4362. 2016. doi:x.2903/j.efsa.2016.4362. ISSN 1831-4732.
- ^ Hillyer, Julián F.; Albrecht, Ralph Thou. (2001). "Gastrointestinal persorption and tissue distribution of differently sized colloidal gold nanoparticles". Journal of Pharmaceutical Sciences. 90 (12): 1927–1936. doi:x.1002/jps.1143. ISSN 0022-3549. PMID 11745751.
- ^ Willhite, Calvin C.; Karyakina, Nataliya A.; Yokel, Robert A.; Yenugadhati, Nagarajkumar; Wisniewski, Thomas M.; Arnold, Ian 1000.F.; Momoli, Franco; Krewski, Daniel (2014-09-xviii). "Systematic review of potential health risks posed by pharmaceutical, occupational and consumer exposures to metal and nanoscale aluminum, aluminum oxides, aluminum hydroxide and its soluble salts". Critical Reviews in Toxicology. 44 (sup4): 1–80. doi:10.3109/10408444.2014.934439. ISSN 1040-8444. PMC4997813. PMID 25233067.
- ^ Mizutani, M.; Mikami, B.; Aibara, S.; Hirose, Yard. (2005). "Construction of aluminium-bound ovotransferrin at 2.15 Å resolution". Acta Crystallographica Section D. 61 (12): 1636–42. doi:10.1107/S090744490503266X. PMID 16301797.
- ^ Ultratrace minerals. Authors: Nielsen, Forrest H. USDA, ARS Source: Modern diet in wellness and disease / editors, Maurice East. Shils ... et al. Baltimore: Williams & Wilkins, c1999., p. 283-303. Issue Engagement: 1999 URI: [2]
- ^ Daumann, Lena J. (25 April 2019). "Essential and Ubiquitous: The Emergence of Lanthanide Metallobiochemistry". Angewandte Chemie International Edition. doi:x.1002/anie.201904090. Retrieved xv June 2019.
- ^ Douglas Trick, "The speed of life", New Scientist, No 2419, 1 November 2003.
- ^ Freitas Jr., Robert A. (1999). Nanomedicine. Landes Bioscience. Tables iii–one & 3–ii. ISBN978-ane-57059-680-3.
- ^ Sender, Ron; Fuchs, Shai; Milo, Ron (2016). "Revised estimates for the number of human and leaner cells in the body". PLOS Biology. 14 (eight): e1002533. bioRxivx.1101/036103. doi:10.1371/journal.pbio.1002533. PMC4991899. PMID 27541692.
- ^ American Academy of Microbiology FAQ: Human being Microbiome Archived 31 Dec 2016 at the Wayback Automobile January 2014
- ^ Judah 50. Rosner for Microbe Magazine, February 2014. Ten Times More than Microbial Cells than Trunk Cells in Humans?
- ^ Gilbert, Jack; Blaser, Martin J.; Caporaso, J. Gregory; Jansson, Janet; Lynch, Susan Five.; Knight, Rob (2018-04-10). "Current agreement of the human microbiome". Nature Medicine. 24 (four): 392–400. doi:10.1038/nm.4517. ISSN 1078-8956. PMC7043356. PMID 29634682.
- ^ Sender R, Fuchs S, Milo R (Jan 2016). "Are We Really Vastly Outnumbered? Revisiting the Ratio of Bacterial to Host Cells in Humans". Cell. 164 (three): 337–xl. doi:x.1016/j.prison cell.2016.01.013. PMID 26824647.
Source: https://en.wikipedia.org/wiki/Composition_of_the_human_body
Posted by: millikenanturing.blogspot.com

0 Response to "What Is The Chemical Makeup Of A Person"
Post a Comment